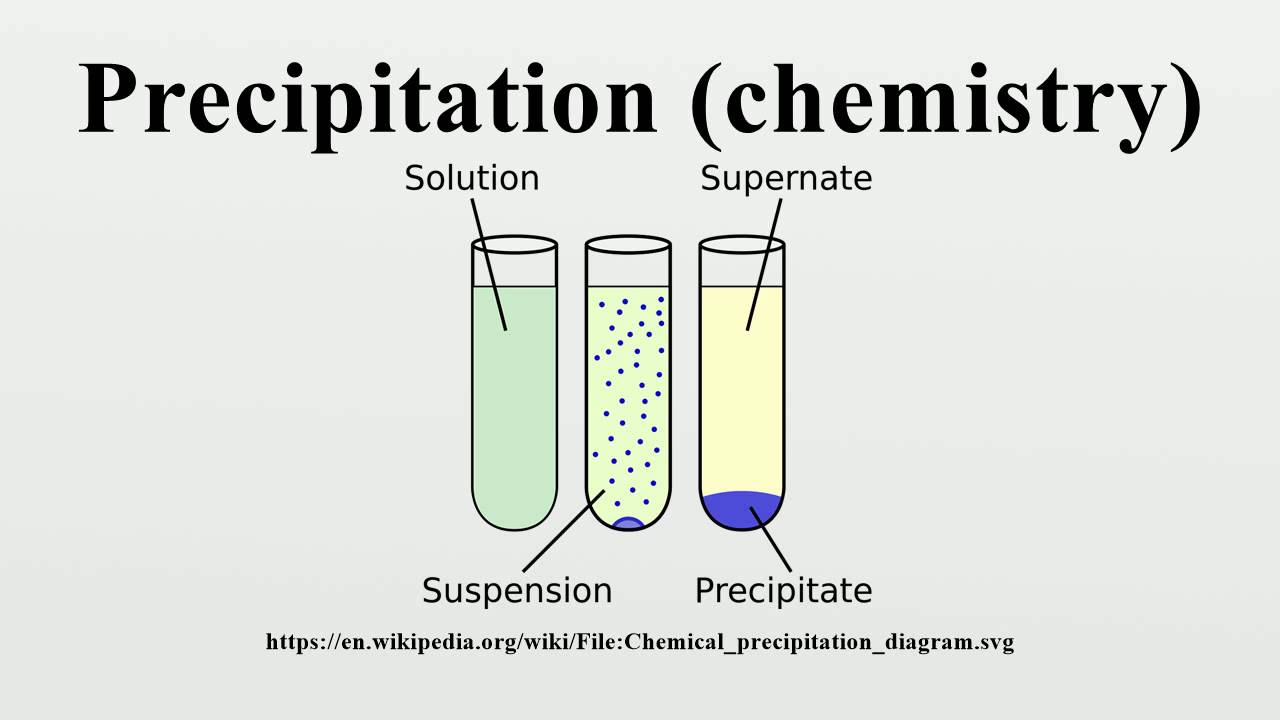
 You can also use it to understand why the body can't absorb minerals present in some foods if an ion such as iron or calcium is "salted out" in the digestive system, the body won't be able to absorb it very efficiently. For example, you can use this trick to remove heavy metal ions from wastewater. It has a number of important applications. It's also called "salting out" a precipitate. Your experiment illustrates the common ion effect: forcing an ionic compound to precipitate from solution by adding one of its ions to the solution. For example, the ion product we calculated in the previous step is less than K sp (4.4 × 10 -6 < 6.5 × 10 -6), so we wouldn't expect any precipitate to form in the tank. If the ion product is less than or equal to the K sp, no precipitate will form. If the ion product is greater than the K sp, the solution is supersaturated, and precipitate will form. For example, if the solution in the tank were 0.010 M Ca(OH) 2 and 0.001 M NaOH, we have:
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
/precipitate-589cb8953df78c47581a9014.jpg)

 RSS Feed
RSS Feed
